Generic vs Brand Name Medications: Are Generic Drugs Really the Same?
Dr. Sarah Mitchell
Certified Pharmacist
When Tom's doctor wrote a prescription for Lipitor, the pharmacist asked if he wanted the generic version, atorvastatin, instead. "It's the same medication, but costs 90% less," she explained. Tom was skeptical. How could it be the same if it costs so much less? Was the quality lower? Would it work as well?
These are questions millions of Americans ask every day. Generic medications can save you thousands of dollars annually, but many people remain uncertain about whether they're truly equivalent to brand-name drugs.
This comprehensive guide explains everything you need to know about generic medications, including how they're regulated, why they cost less, and whether they work as well as brand names.
What Are Generic Medications?
A generic medication is a drug that contains the same active ingredient as a brand-name medication, in the same dosage and form, intended for the same use.
The Brand Name Period
When a pharmaceutical company develops a new drug, they patent it, giving them exclusive rights to sell it for typically 20 years (from patent filing). During this period, no one else can manufacture the drug. The company sets prices to recoup research and development costs plus profit.
Generic Entry
When patents expire, other manufacturers can apply to make generic versions. These generics must prove they're equivalent to the brand-name drug. Once approved, multiple companies often manufacture the same generic, creating price competition.
Legal Definition
According to the FDA, a generic drug is identical - or bioequivalent - to a brand-name drug in dosage form, safety, strength, route of administration, quality, performance characteristics, and intended use.
How Generic Drugs Are Regulated
Generic drugs face strict FDA approval requirements.
FDA Approval Process
Generic manufacturers must prove their drug is bioequivalent to the brand-name version. This means the generic: has the same active ingredient, is the same strength, is taken the same way (oral, injection, etc.), meets the same quality standards, and is intended for the same use.
Bioequivalence Testing
The FDA requires studies showing the generic drug: reaches the bloodstream at the same rate and to the same extent as the brand-name drug. This is measured through blood tests comparing how much active ingredient enters the bloodstream and how long it takes.
The FDA allows a small range of difference (typically 80-125%), but studies show generics usually fall within 3-4% of the brand name - well within this range.
Manufacturing Standards
Generic drug manufacturers must meet the same strict manufacturing standards as brand-name manufacturers. FDA inspects generic drug manufacturing facilities just like brand-name facilities. Generics must meet the same batch-to-batch consistency requirements.
Ongoing Monitoring
FDA continues to monitor generic drugs after approval through inspections, testing, and adverse event reporting. If quality issues arise, FDA can recall generic medications just like brand names.
What's the Same in Generic Drugs?
Understanding what must be identical helps build confidence in generics.
Always Identical
Active ingredient: The chemical that produces the therapeutic effect. Dosage strength: Amount of active ingredient per pill/dose. Dosage form: Tablet, capsule, liquid, etc. Route of administration: Oral, injection, topical, etc. Safety profile: Same warnings, precautions, and contraindications. Effectiveness: Must work the same way in the body. Quality: Must meet same pharmaceutical standards. Manufacturing standards: Same FDA requirements.
These are not negotiable - they must be the same for FDA approval.
What Can Be Different in Generic Drugs?
Some differences are allowed and don't affect how the drug works.
May Be Different
Inactive ingredients: Fillers, binders, colors, and flavors can differ. Shape and color: Appearance can be different. Packaging: Bottle style, labels (beyond required information). Scoring: Whether tablets can be split. Manufacturer: Different company makes it. Price: Usually much less expensive.
Why Inactive Ingredients Matter
In most cases, different inactive ingredients don't affect how the drug works. However, some people may: be allergic to specific inactive ingredients, notice different tablet coating affecting absorption slightly, or have sensitivity to different dyes or fillers.
These situations are rare but possible.
Why Generic Drugs Cost So Much Less
If they're the same, why the huge price difference?
No Research and Development Costs
Brand-name companies spend $1-3 billion developing each new drug. This includes years of research, clinical trials, and failed attempts. Generic manufacturers skip this - they just copy the proven drug.
No Marketing Costs
Brand-name companies spend billions on marketing and advertising. Generic manufacturers spend minimal amounts on marketing. You've seen Cialis commercials; you've never seen generic tadalafil commercials.
Competition
Once a drug goes generic, multiple companies can manufacture it. 10-20 generic manufacturers might compete for the same drug. Competition drives prices down dramatically.
Faster FDA Approval
Generic approval process is abbreviated compared to new drugs. Takes months instead of years. Lower approval costs.
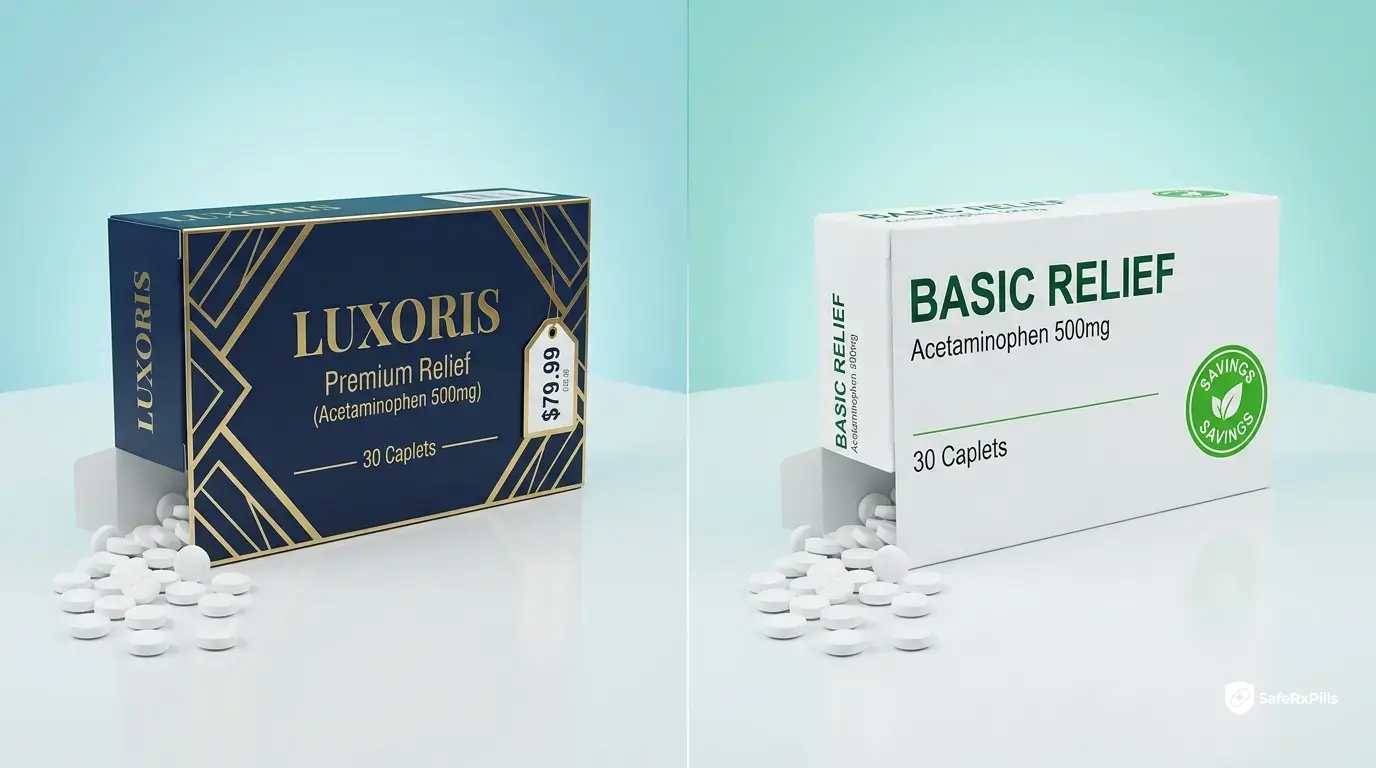
Example Price Comparisons
Lipitor (atorvastatin): Brand: $400-500 for 30 pills. Generic: $10-20 for 30 pills.
Viagra (sildenafil): Brand: $70-90 per pill. Generic: $2-8 per pill.
Prozac (fluoxetine): Brand: $200-300 for 30 pills. Generic: $4-10 for 30 pills.
These are typical savings of 80-95%.
Do Generic Drugs Work as Well?
The evidence is clear: yes, for the vast majority of medications.
Clinical Evidence
FDA studies of thousands of generic drugs show they work as well as brand names. Average difference in bioavailability: 3.5% (well within the 20% allowed range). Patient outcomes are equivalent in clinical studies.
When Generics Work Identically
For most medications, including antibiotics, blood pressure medications, cholesterol drugs, antidepressants, diabetes medications, and pain relievers, generics work identically to brand names.
Narrow Therapeutic Index Drugs
A small category of drugs requires extra scrutiny - these have a narrow therapeutic index, meaning small differences in blood levels can affect effectiveness or cause side effects.
Examples include: Warfarin (blood thinner), Levothyroxine (thyroid medication), Seizure medications, and some heart medications.
For these drugs, FDA has stricter requirements, and some doctors prefer patients stay on one consistent version (brand or generic from the same manufacturer).
Patient Perceptions
Studies show that when patients don't know they're taking a generic, they report the same effectiveness and side effects as brand names. When patients know they're taking a generic, some report "differences" - likely due to expectations, not actual drug effects.
This is the placebo/nocebo effect at work.
Common Myths About Generic Drugs
Let's address misconceptions.
Myth 1: "Generic drugs are lower quality"
Truth: Generics must meet the same FDA quality standards. Same purity requirements, same strength specifications, same manufacturing standards. FDA inspects generic facilities just like brand-name facilities.
Myth 2: "Generic drugs are made in less regulated facilities"
Truth: Generic drug facilities worldwide must meet FDA standards. FDA inspects foreign and domestic generic facilities regularly. Non-compliant facilities are shut down regardless of location.
Myth 3: "Generic drugs don't work as well"
Truth: FDA requires proof of bioequivalence. Clinical studies show equivalent outcomes. Any difference is typically 3-4%, not clinically significant.
Myth 4: "Brand names are safer"
Truth: Generics have the same safety profile. Same active ingredient means same potential side effects. Same warning labels and precautions.
Myth 5: "Doctors prefer brand names"
Truth: Most doctors routinely prescribe generics. Insurance companies and healthcare systems prefer generics. Some doctors specify brand only for narrow therapeutic index drugs.
Myth 6: "I can tell the difference in effectiveness"
Truth: Properly blinded studies show patients can't identify generic vs brand. Perceived differences are usually placebo effect or variations in disease itself.
When to Choose Generic vs Brand Name
For most people, most of the time, generic is the right choice.
Choose Generic When
You want to save money (80-95% savings). Your medication is not a narrow therapeutic index drug. Your doctor doesn't specifically require brand name. You're taking long-term maintenance medications. You have high medication costs.
This applies to the vast majority of prescriptions.
Consider Brand Name When
You have narrow therapeutic index drug (warfarin, thyroid medication, seizure meds). You've tried generic and experienced genuine issues (not just worry). Your doctor specifically recommends brand for medical reasons. Cost difference is minimal with your insurance. You're switching between multiple generic manufacturers frequently.
The Insurance Factor
Most insurance plans heavily favor generics with much lower copays. Brand name copays might be $50-100 while generic copays are $5-15. Some plans won't cover brand names if a generic exists. Check your insurance formulary.
How to Switch from Brand to Generic
If you're currently taking a brand-name medication, switching to generic is usually simple.
Talk to Your Doctor
Discuss the switch - most doctors support it. For narrow therapeutic index drugs, ask if monitoring is needed. Get confirmation the generic is appropriate for you.
At the Pharmacy
Ask pharmacist about the generic manufacturer. For narrow therapeutic index drugs, ask them to note the manufacturer so refills come from the same one. Verify the generic is FDA-approved.
Monitor Your Response
For the first month after switching, pay attention to effectiveness and side effects. Keep a journal if you're concerned. Report any genuine concerns to your doctor.
For Narrow Therapeutic Index Drugs
May need blood tests to confirm therapeutic levels. Stay consistent with one generic manufacturer. Inform all healthcare providers you're taking generic.
Generic Drug Quality Issues: Reality Check
While generic drugs are generally high quality, issues occasionally arise.
Real Quality Concerns
Some foreign manufacturers have been cited for quality violations. FDA has increased inspections and recalls when needed. A few generics have been recalled for quality issues.
Perspective
Brand-name drugs are also recalled for quality issues. Overall recall rate is similar for generics and brands. When problems occur, FDA acts quickly.
What You Can Do
Buy from reputable pharmacies. Report unusual side effects or effectiveness issues. If concerned, ask pharmacist about manufacturer. Stay informed about FDA recalls.
Saving Money with Generic Drugs
Maximize your savings with these strategies.
Ask Your Doctor to Prescribe Generics
Request generic prescriptions when available. Ask doctor to avoid "dispense as written" designation. Discuss any concerns about specific medications.
Check if a Generic Exists
Ask your pharmacist if a generic version is available. Some newer medications don't have generics yet (still under patent). Generics typically become available 20 years after drug's patent filing.
Shop Around
Generic prices vary significantly between pharmacies. Use GoodRx or similar tools to compare. Consider mail-order pharmacies. Ask about 90-day supplies (often cheaper per pill).
Don't Assume Brand is Better
Unless your doctor has specific medical reasons, generic is appropriate. Paying more doesn't mean better quality. Trust the FDA approval process.
The Future of Generic Drugs
Biosimilars
New category: biosimilars are versions of biologic drugs (made from living cells). Similar to generics but for complex biologic medications. Will provide savings on expensive cancer, arthritis, and other specialty medications.
Increasing Generic Competition
More manufacturers entering generic market. Faster FDA approval for generics in shortage. Policies encouraging generic competition.
Patient Education
Healthcare systems increasingly educating patients about generic equivalence. Reducing stigma and misconceptions. Emphasizing cost savings for healthcare system and patients.
The Bottom Line
Generic medications are equivalent to brand-name drugs in every way that matters: same active ingredient, same strength, same effectiveness, same safety profile. They cost 80-95% less because manufacturers avoid research, development, and marketing costs.
Key points to remember: FDA requires proof generics work the same as brand names, quality standards are identical for generics and brands, billions of prescriptions prove generic safety and effectiveness, you can save thousands of dollars annually with generics, for most medications choosing generic is a smart decision, and rare exceptions exist for narrow therapeutic index drugs.
The evidence is overwhelming: generic drugs are a safe, effective way to access the medications you need while saving significant money. Don't let myths prevent you from benefiting from generic medications.
Talk to your doctor and pharmacist. Ask questions. Make informed decisions. In the vast majority of cases, generic is the right choice.
Dr. Sarah Mitchell
PharmD, Clinical Pharmacist
Experienced pharmacist and healthcare writer specializing in medication safety and patient education.
Comments (0)
Leave a Comment
No comments yet. Be the first to share your thoughts!
Related Articles

How to Save Up to 80% on Prescription Medications: A Practical Guide for Americans
April 30, 2026

How to Order Medications Online Safely: Complete Guide to Legitimate Online Pharmacies
April 30, 2026
